Introduction
Most institutions do not struggle with COE accreditation because they lack effort. They struggle because compliance is managed informally. Evidence sits in scattered folders, version control is inconsistent, and operational knowledge changes depending on who is working that day. In a COE environment, that creates real risk because accreditation decisions depend on written documentation, not verbal explanations, assumptions, or institutional memory.
A COE Quality Management System (QMS) is how an institution replaces reactive compliance with a controlled, repeatable operating model. Instead of relying on last-minute document gathering, a COE compliance system defines ownership, controls evidence, schedules monitoring, documents corrective action, and keeps the accreditation record current year-round.
For institutions working through institutional accreditation, the real question is not whether staff care about compliance. The real question is whether the institution can produce accurate, organized, and defensible documentation when COE asks for it.
For institutions operating in the United States, COE accreditation compliance is not only a regulatory requirement but also a critical factor in maintaining eligibility, operational stability, and public trust. U.S.-based institutions must align their COE Quality Management System (QMS) with federal expectations, state authorization requirements, and accreditor-specific documentation standards to remain compliant and audit-ready.
This article explains how to build a practical COE Quality Management System (QMS) that supports accreditation compliance, evidence control, EDvera reporting, and continuous readiness.
Quick Answer
A COE Quality Management System (QMS) is a year-round compliance framework that connects COE accreditation requirements to clear owners, controlled documentation, routine monitoring, and documented corrective action. A strong system includes a permanent accreditation file, annual report readiness, internal audits, public disclosure review, and leadership oversight. The goal is simple: produce credible written documentation on demand and reduce accreditation risk before it becomes a finding.
Key Takeaways
COE accreditation is documentation-driven, which means institutions need proof, not explanations. A practical COE QMS must maintain a permanent accreditation file, control official records, support the annual report process in EDvera, and document corrective action with follow-up checks. Institutions that manage COE compliance as an ongoing operating condition, rather than a once-a-year event, are in a much stronger position during reviews, reporting cycles, and substantive change activity.
What a COE Quality Management System (QMS) Means in Accreditation Compliance
In plain English, a COE Quality Management System is the structure that allows your institution to answer the same compliance questions consistently every time. Who owns the requirement? What process supports it? Where is the evidence stored? How often is it reviewed? What happens when a gap is identified?
That is the difference between a real accreditation compliance system and a collection of disconnected documents. In a COE environment, documentation quality is not a side issue. It is part of institutional credibility. If files are incomplete, contradictory, outdated, or impossible to retrieve, the institution appears uncontrolled even when the underlying operations may be sound.
A strong QMS also supports the broader accreditation process. It creates a stable written record across reporting cycles, self-studies, team visits, annual submissions, and institutional responses. Without that structure, institutions often spend more time reconstructing compliance than managing it.
Key Terms in a COE Quality Management System
A COE Quality Management System (QMS) is the full set of processes, controls, review cycles, and improvement actions used to maintain accreditation compliance. It is not a binder, a one-time project, or a vague quality initiative.
A permanent accreditation file is the controlled institutional record that preserves essential accreditation history in an organized and retrievable format. This file should function as a living system, not a storage closet for old PDFs.
Evidence control refers to the rules that govern document approval, versioning, storage, naming conventions, access, and retrieval. If two departments produce different versions of the same policy, evidence control has failed.
Corrective action means addressing the cause of a compliance gap, not just fixing the visible symptom. A real corrective action record includes the issue, cause, action taken, proof of implementation, and follow-up verification.
EDvera annual report readiness means the institution can pull accurate information from controlled records and complete the reporting cycle without last-minute scrambling.
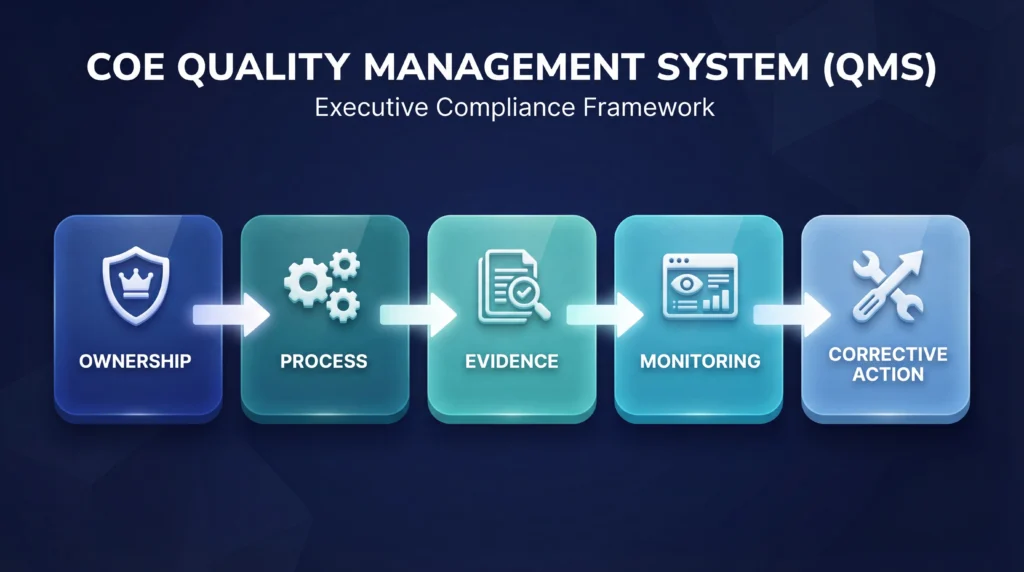
The AEC Practical COE Quality Management System Framework

A practical COE QMS works best when it is built on a few durable controls rather than an oversized compliance bureaucracy. Institutions do not need fifty templates. They need system discipline.
The first control is a COE standards crosswalk. This crosswalk should connect each requirement area to a responsible owner, operational process, evidence source, and monitoring cadence. Shared responsibility sounds collaborative until no one is actually accountable.
The second control is evidence control. Official records should have one location of record, a clear owner, a current approved version, and a defined retention logic. This is how institutions reduce contradictions, outdated submissions, and staff confusion.
The third control is permanent accreditation file discipline. Your institution should know exactly what belongs in that file, who updates it, when it is reviewed, and how completeness is checked. The file should be part of governance, not an afterthought.
The fourth control is annual report readiness. If an institution waits until reporting season to figure out where data lives, the system is already broken.
The fifth control is leadership oversight. Senior leadership should review compliance status, deadlines, open findings, corrective actions, and documentation risks on a scheduled basis. That is how institutions demonstrate institutional control instead of informal dependency on a few knowledgeable employees.
Step-by-Step: How to Build a COE Quality Management System (QMS)
Step 1: Define scope and institutional obligations
Start by defining the actual scope of your COE compliance system. That includes campuses, instructional modalities, programs, reporting responsibilities, public disclosures, and any regulatory overlap that affects documentation. If your operations have changed but your compliance map has not, your QMS is already behind.
Step 2: Build the COE standards crosswalk
Before you clean files, define what those files are supposed to prove. A standards crosswalk should connect each COE requirement to the responsible owner, operational process, evidence artifact, and review schedule. This step prevents random file cleanup from being mistaken for accreditation readiness.
Step 3: Establish evidence control rules
Create practical evidence control rules that staff can actually follow. Define where official records live, how files are named, who approves updates, how versions are replaced, and what counts as the official submission copy. If documentation practices are too complicated, staff will ignore them.
Step 4: Stand up the permanent accreditation file
The permanent accreditation file should be treated as a controlled institutional system. Assign ownership. Set a review schedule. Use chronological organization. Confirm required items are complete and current. This is where many institutions look stronger or weaker than they really are.
Step 5: Align annual reporting with the operating calendar
Annual reporting should be built into the institution’s annual compliance calendar, not treated as an emergency project. EDvera reporting works best when data and supporting documents are maintained throughout the year. That turns the annual report into a validation step instead of a scramble.
Step 6: Implement internal monitoring and internal audits
Monitoring checks whether compliance processes are running consistently. Internal audits test whether they can withstand scrutiny. Both should be documented. If the institution discovers issues early, corrective action is cheaper, cleaner, and more credible.
Step 7: Operationalize corrective action with proof
Corrective action should be managed like a governance item. Every issue should have an owner, due date, documented action, implementation evidence, and follow-up review. Institutions that only document the fix, but not the control change behind the fix, usually repeat the same problem later.
COE Evidence Control, EDvera Reporting, and Documentation Requirements
A COE compliance system depends on written evidence that is accurate, current, and retrievable. That means evidence control is not administrative housekeeping. It is part of the institution’s accreditation infrastructure.
Your QMS should define who can prepare records, who can approve them, who can submit through EDvera, and where the institution retains its own final copies. Platform access alone is not a records strategy. Institutions still need internal control over the written record.
Evidence control should also extend to public-facing materials. Accreditation status language on the website, catalog, and marketing materials should be reviewed on a schedule. Institutions can create avoidable compliance risk when public disclosures drift away from official status language.
This is also the right place to connect documentation discipline to the self-study. A cleaner evidence system makes self-study preparation faster, more consistent, and less dependent on last-minute reconstruction.
Monitoring, Internal Audits, and Corrective Action in COE Compliance
Monitoring is the routine practice of checking whether required processes are functioning and whether evidence is being generated consistently. Internal audits go deeper. They test whether the compliance system can actually defend the institution when COE asks questions.
A mature COE Quality Management System uses both. Monitoring identifies drift early. Internal audits evaluate whether the system is still aligned with requirements, deadlines, and actual operations.
Corrective action should never be reduced to a one-line note saying the issue was fixed. Strong accreditation documentation shows the original gap, the reason it happened, the action taken, the proof of implementation, and the effectiveness check after the change. That level of written discipline is what separates operational maturity from hopeful storytelling.
Common COE Compliance Mistakes in QMS Implementation
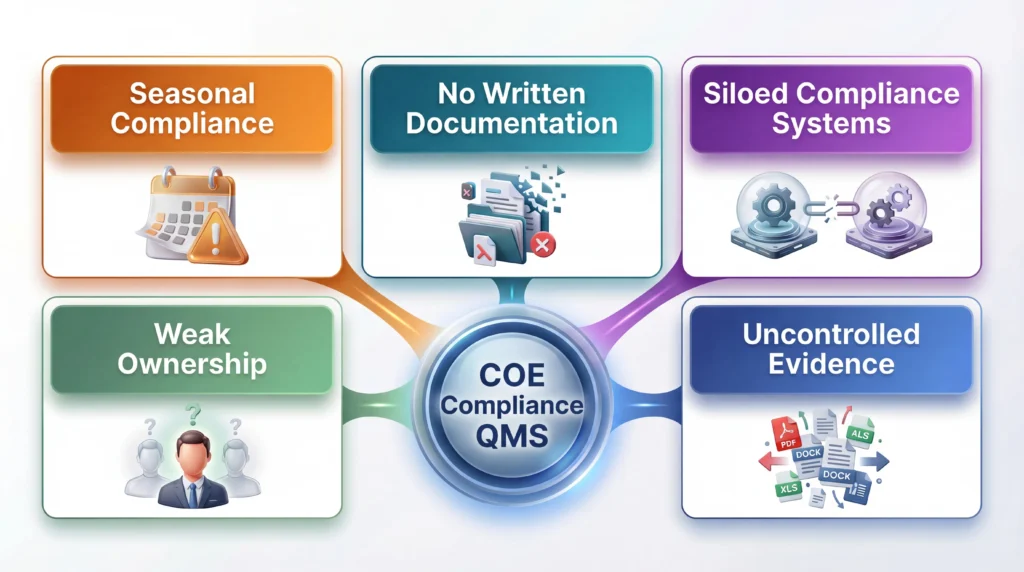
The first mistake is treating COE compliance as a seasonal project tied to the annual report or a site visit. That mindset creates rushed documentation, weak review cycles, and recurring errors.
The second mistake is relying on verbal explanations, email trails, or unwritten practices instead of controlled documentation. When the institution cannot produce a stable written record, the explanation usually does not matter.
The third mistake is weak ownership. If responsibility belongs to “the team” or “whoever handled it last year,” accountability disappears and documentation quality slips.
The fourth mistake is poor evidence control. Multiple versions, conflicting dates, unclear approvals, and scattered storage systems make even strong institutions look unreliable.
The fifth mistake is separating accreditation from state approval. In real institutional operations, those obligations often overlap. If timelines, disclosures, or program changes are tracked in separate silos, one compliance problem can easily create another.
What COE Reviewers Look For
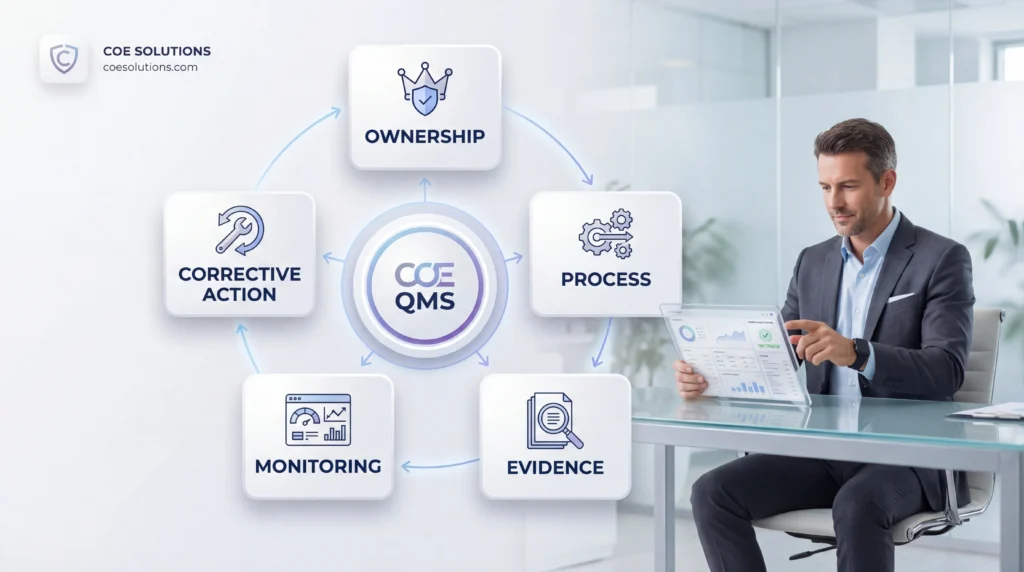
COE reviewers are ultimately looking for institutional control. They want to see that the institution can consistently meet requirements and demonstrate that through a reliable written record.
That means reviewers look for clear ownership, coherent processes, retrievable documentation, timely reporting, and a visible relationship between monitoring and corrective action. They are not just reading policies. They are evaluating whether the institution operates in a way that supports those policies.
A well-run COE Quality Management System makes that visible. It shows that the institution governs compliance deliberately, rather than relying on memory, personalities, or deadline pressure.
COE Accreditation and State Authorization: What Institutions Must Align
Even when an institution is focused on COE accreditation, it still has to manage other legal and regulatory obligations that affect operations, disclosures, programs, and timing. For many institutions, the biggest overlap is between accreditation readiness and state approval requirements.
That is why the QMS should track state actions alongside accreditation actions. New programs, location changes, modality shifts, catalog revisions, and public claims can all create parallel obligations. Institutions that ignore this overlap often discover the conflict too late.
A strong COE compliance system does not treat accreditation and state obligations as separate universes. It treats them as related control points within one operating environment.
FAQ
What are COE accreditation documentation requirements?
COE accreditation requirements are documentation-driven, meaning institutions must provide written evidence that demonstrates compliance with COE standards, criteria, and policies. This includes controlled policies, operational records, monitoring results, and supporting documentation that can be reviewed and verified during accreditation evaluations or reporting cycles.
What is included in a COE permanent accreditation file?
A COE permanent accreditation file is a structured, chronological record of institutional accreditation documentation. It typically includes correspondence with COE, the most recent self-study, team reports, institutional responses, substantive change applications, and annual reports with supporting documentation. The file must be complete, organized, and retrievable at all times.
When is the COE annual report due in EDvera?
The COE annual report is typically due by December 15 each year and is submitted electronically through EDvera. Once submitted, the report is locked and cannot be modified, which makes early preparation and controlled documentation essential for accurate reporting.
What happens if an institution misses a COE reporting deadline?
Missing a COE reporting deadline can create significant compliance risk. Institutions may face findings, additional reporting requirements, or more serious accreditation consequences depending on the situation. A strong COE Quality Management System (QMS) ensures deadlines are tracked, managed, and supported by controlled documentation to prevent missed submissions.
How do institutions prepare for a COE accreditation review?
Institutions prepare for a COE accreditation review by implementing a structured compliance system that includes clear ownership, controlled evidence, routine monitoring, internal audits, and documented corrective action. Preparation is not a one-time activity but an ongoing process supported by a well-designed COE Quality Management System (QMS).
Next Steps
If you want COE readiness to be predictable, build the system that makes it predictable. Start with your standards crosswalk, evidence control rules, permanent accreditation file, annual reporting calendar, and internal audit schedule. Then make leadership review part of the operating rhythm.
Institutions that want outside support in designing or rebuilding a documentation-driven compliance system can work with AEC for higher education accreditation consulting focused on accreditation readiness, evidence control, corrective action planning, and practical implementation.
About the Author
Accreditation Expert Consulting (AEC) provides fact-based guidance on accreditation readiness, compliance systems, documentation control, corrective action planning, and institutional evidence management for higher education organizations operating in complex regulatory environments.

This article is provided for general informational purposes and does not constitute legal advice. COE requirements, manuals, reporting workflows, and related obligations should always be confirmed through current official COE resources and applicable regulatory sources.



