
How to Investigate Accreditation Non-Compliance
Introduction
When an accreditor flags accreditation non-compliance, most institutions do the same predictable thing: they rush to “fix it,” start drafting a response, and hope the wording sounds confident enough to make the problem go away. That approach usually produces a glossy narrative and a weak outcome, because reviewers aren’t grading your tone. They’re grading your evidence, your controls, and whether your institution can reliably meet requirements without last-minute heroics.
Accreditation non-compliance is rarely just a single broken item. It’s usually a signal that something in your institutional system failed to produce consistent, verifiable compliance. Sometimes it’s performance. Often, it’s documentation and evidence management. Frequently it’s ownership and governance, meaning nobody was truly accountable for the requirement, and the institution didn’t catch the gap before the reviewer did.
Investigating institutional accreditation non-compliance findings is a skill. The goal isn’t to sound impressive. The goal is to be correct, and to be provably correct. That means turning a finding into a clear, requirement-based problem statement, confirming scope and evidence, identifying root cause, and implementing corrective actions that create proof and reduce the chance of the same issue coming back under a different label next year. This article walks you through a practical, accreditation-ready method that reviewers can follow, verify, and trust.
Quick Answer
Investigating accreditation non-compliance involves a structured process that connects the reviewer’s finding to a clear corrective action and long-term compliance control.
A typical investigation includes the following steps:
- Translate the finding into a requirement-based problem statement tied to the exact accreditation standard.
- Confirm scope and supporting evidence, including programs, campuses, modalities, and student populations affected.
- Separate symptoms from root causes using structured root cause analysis.
- Design corrective actions that address the underlying cause rather than just fixing the immediate issue.
- Assign accountable owners, deadlines, and verification evidence for each corrective action.
- Conduct an effectiveness check over time to confirm the corrective action actually prevents recurrence.
The objective is not simply writing a persuasive response. The objective is building a system that consistently produces compliance and can demonstrate it with verifiable evidence whenever reviewers ask.
Key Takeaways
Strong responses to accreditation non-compliance findings are built on evidence, not persuasion.
- Treat the finding as a testable claim tied to a requirement, not a personal critique.
- Separate the visible symptom from the root cause that created it.
- Ensure corrective actions change the system, not just patch the immediate incident.
- Assign clear ownership, deadlines, and retrievable evidence for every corrective action.
- Include an effectiveness check so the institution can demonstrate that the fix actually worked over time.
What Non-Compliance Means in Accreditation
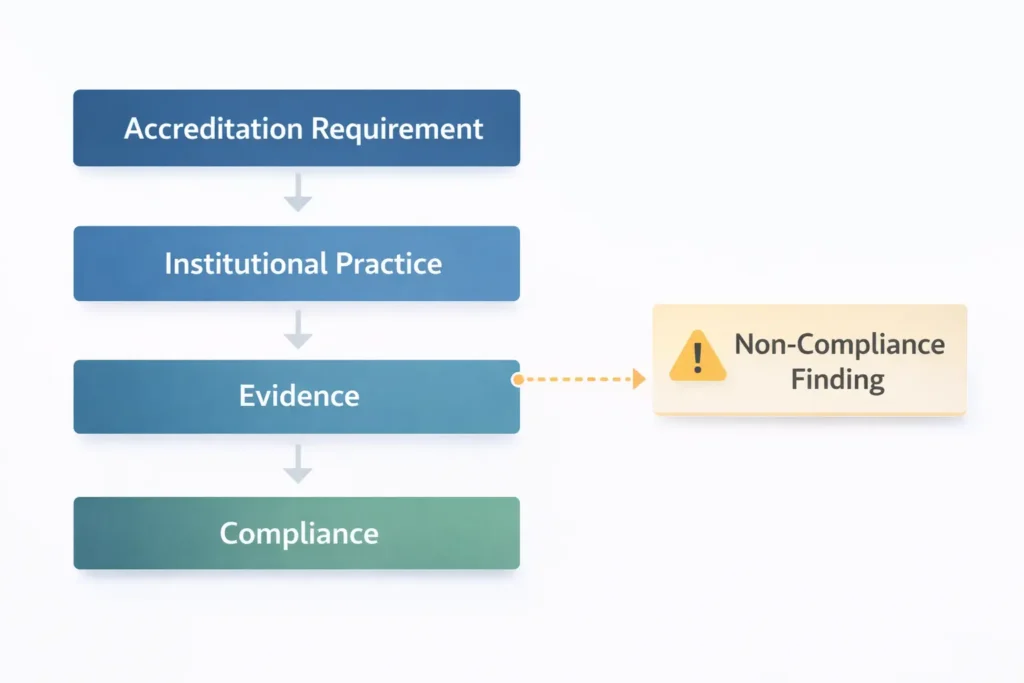
In higher education accreditation process settings, non-compliance often appears as a finding, deficiency, citation, or “standard not met,” depending on the accreditor or regulatory body. While the terminology may differ, the underlying logic is consistent: the institution did not demonstrate that it meets a specific requirement. Accreditation systems are designed to evaluate whether institutions can consistently meet defined quality standards and provide verifiable evidence of compliance. As explained by the Council for Higher Education Accreditation (CHEA), accreditation functions as a formal quality assurance process through which institutions are reviewed against established standards and expected to demonstrate ongoing compliance.
This gap can occur for several reasons. In some cases, institutional practice does not fully align with the requirement. In others, the practice may exist but the supporting evidence is incomplete, inconsistent, or difficult to verify. Non-compliance can also arise when implementation varies across programs, campuses, locations, delivery modalities, or student populations.
A critical mindset shift is necessary when responding to accreditation findings. Institutions should treat a finding as a claim that must be evaluated and tested against evidence tied to the requirement. When teams treat the issue as a debate about wording or interpretation, responses often drift into defensiveness and narrative explanations.
Accreditors and reviewers are not evaluating tone or intent. They are evaluating whether the institution can consistently meet requirements and provide verifiable evidence of compliance without last-minute reconstruction of documentation.
Key Terms and Definitions
Non-compliance: A documented gap where institutional practice or evidence does not meet an accreditation requirement, standard, policy expectation, or regulatory obligation.
Finding: A formal statement issued by reviewers indicating that a requirement is not met or not sufficiently demonstrated, usually referencing both the requirement and the evidence reviewed.
Correction: An immediate action taken to resolve a specific instance of non-compliance, such as updating a disclosure, correcting an error, or completing missing documentation.
Corrective Action: A systemic change designed to eliminate the underlying cause of non-compliance and prevent recurrence. Examples include clarifying ownership, redesigning workflow controls, strengthening governance oversight, or implementing ongoing monitoring processes.
Root Cause: The underlying factor or set of factors that allowed the non-compliance to occur. Root causes often relate to governance structures, process design, training controls, accountability gaps, resource limitations, or insufficient monitoring.
Evidence: Verifiable documentation or data demonstrating compliance. Effective evidence clearly shows what was done, when it occurred, and what outcomes were produced.
Effectiveness Check: A planned process used to confirm that corrective actions resolved the issue and reduced the risk of recurrence over time. This may involve internal audits, monitoring reports, performance indicators, or compliance reviews.
Common Causes of Accreditation Non-Compliance

Accreditation non-compliance findings rarely happen because of a single mistake. In most cases, they reflect weaknesses in institutional systems that prevent consistent documentation, oversight, or implementation of accreditation requirements. Many institutions address these challenges through structured accreditation compliance frameworks and external advisory support designed to align institutional processes with accreditation standards. Federal oversight of recognized accrediting agencies in the United States also emphasizes the importance of consistent institutional compliance systems, as described by the U.S. Department of Education.
Across many accreditation reviews, several recurring causes appear repeatedly.
Unclear ownership of compliance responsibilities
When no specific role is accountable for a requirement, tasks such as policy reviews, reporting, or documentation updates are often delayed or overlooked.
Weak governance or oversight structures
Institutions sometimes lack consistent monitoring from compliance committees, academic leadership, or internal review bodies. Without oversight, small compliance gaps can persist for long periods.
Inconsistent documentation and evidence practices
Evidence may exist but be scattered across departments, stored in different systems, or difficult to retrieve during a review.
Lack of monitoring or internal audit processes
Without periodic internal checks, institutions often discover compliance issues only when an external reviewer identifies them.
Misalignment between policy and operational practice
Policies may describe compliant processes, but daily operational practices may not fully reflect those policies.
Understanding these underlying causes is essential before designing corrective actions. If an institution focuses only on the visible symptom of a finding, the same issue often reappears later under a different standard or requirement.
The AEC 9-Step Method for Investigating Accreditation Non-Compliance (2026)
A credible response to accreditation non-compliance findings starts with a disciplined investigation. The method below works across accreditors and regulatory environments because it follows a universal logic: requirement, evidence, cause, action, proof, and effectiveness.
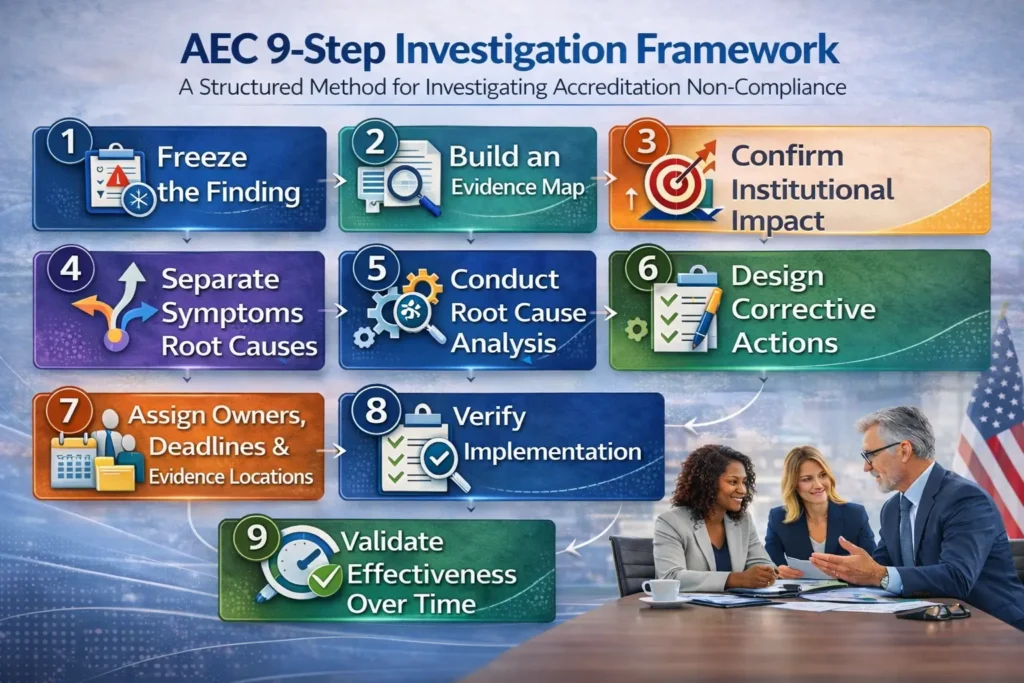
The difference between institutions that successfully resolve accreditation findings and those that repeatedly face the same issues is often the investigation process itself. Some institutions treat the finding like a writing exercise, focusing on narrative responses. Others treat it like a controlled compliance investigation designed to identify root causes and implement lasting corrective actions.
The AEC 9-step framework below provides a practical structure for investigating accreditation non-compliance and building defensible corrective action responses.
Step 1: Freeze the Finding Before You “Fix” Anything
Start by capturing the accreditation non-compliance finding exactly as written and anchoring it to the requirement it references. Document the cited standard or regulation, the finding language verbatim, the scope (program, campus, modality, or student population), the relevant time period, and the evidence reviewers referenced.
This prevents a common institutional failure where different departments begin addressing different interpretations of the same problem, resulting in inconsistent responses.
This step also clarifies the type of gap involved. Some findings primarily reflect evidence gaps, others indicate implementation gaps, and some represent actual performance failures. The corrective response must match the type of gap involved.
Step 2: Build an Evidence Map
Separate the available documentation into three categories:
- evidence reviewers examined
- evidence that exists but was not clearly presented
- evidence that does not exist
Many accreditation findings occur not because institutions lack compliant practices, but because documentation is scattered across departments, inconsistently labeled, not version-controlled, or difficult to retrieve during review.
From a reviewer’s perspective, “we have it somewhere” is equivalent to not having it at all.
By the end of this step, the investigation should clearly identify whether the issue involves:
- missing documentation
- inconsistent implementation
- lack of institutional controls
Step 3: Confirm and Document Institutional Impact
Not every accreditation non-compliance carries the same level of institutional risk. Treating all findings as equally severe can reduce the credibility of the response.
Reviewers and regulators typically focus more heavily on gaps affecting:
- student protections
- academic integrity
- learning outcomes
- governance
- financial and administrative oversight
A strong impact assessment answers three questions:
- What was affected?
- Who was affected?
- What evidence demonstrates the impact?
Effective impact statements rely on records, audit logs, outcome data, and documented processes, rather than assumptions or internal opinions.
Step 4: Separate Symptoms from Root Causes
Symptoms describe what happened. Root causes explain why it happened.
Many institutions respond only to symptoms, which leads to repeated findings in future accreditation cycles.
Examples include:
- An outdated policy may actually reflect missing ownership and review cycles
- Inconsistent documentation may indicate weak evidence governance
- Staff non-compliance may reflect unclear procedures or missing monitoring controls
Reviewers can quickly recognize when institutions address only the symptom while leaving the underlying system unchanged.
Step 5: Conduct Root Cause Analysis
Root cause analysis should produce evidence-based conclusions, not brainstorming summaries.
The objective is to identify the small number of systemic factors responsible for most of the compliance failure.
Common root causes in accreditation investigations include:
- unclear ownership of compliance responsibilities
- weak governance oversight
- inconsistent institutional processes
- inadequate training controls
- flaws in the 4 stages of curriculum development
- system or tool limitations
- insufficient staffing or resources
- missing monitoring and internal audits
Each identified root cause should be supported by documented evidence, observed patterns, or audit results.
Step 6: Design System-Level Corrective Actions
Corrective actions must address both the requirement and the root cause.
For example:
- If the root cause is unclear ownership, the corrective action should formalize responsibility and oversight.
- If the root cause is inconsistent practice across units, the corrective action should standardize procedures and introduce monitoring.
- If the root cause is evidence management failure, the corrective action should establish documentation controls such as retention rules, version control, and evidence indexing.
Weak corrective actions rely on individual effort or memory. Strong corrective actions create institutional systems that consistently generate compliance evidence.
Step 7: Assign Owners, Deadlines, and Evidence Locations
Every corrective action must include:
- a clearly accountable role (not simply a committee)
- a defined completion deadline
- specific deliverables
- a designated location where documentation and evidence will be stored
Reviewers frequently test institutional readiness by asking for supporting documentation. Institutions that cannot retrieve evidence quickly often lose credibility.
This step also clarifies completion criteria. For example, “policy updated” is not complete until the policy is approved, published, communicated, implemented, and monitored.
Step 8: Verify Implementation
Verification answers a simple but critical question:
Did the institution actually implement the corrective actions it promised?
Verification evidence should be:
- dated
- retrievable
- clearly linked to the corrective action
Typical verification evidence includes:
- approved policies with version history
- governance meeting minutes
- controlled training completion records
- updated catalogs or website disclosures
- internal audit logs
- system configuration documentation
The structure and organization of evidence can significantly influence reviewer confidence.
Step 9: Validate Effectiveness Over Time
Implementation alone does not guarantee long-term compliance.
Effectiveness validation confirms whether the corrective action actually reduced the risk of recurrence.
Common effectiveness checks include:
- periodic internal audits
- document or file reviews
- KPI monitoring
- compliance committee oversight reviews
If the corrective action is new and long-term monitoring results are not yet available, institutions should provide implementation evidence and clearly define the planned effectiveness monitoring process.
Transparency in this stage often builds greater credibility than overconfident claims.
The Non-Compliance Investigation Brief

Before drafting any formal response to an accreditation non-compliance finding, institutions should prepare a concise investigation brief. A one-page investigation brief helps align teams, maintain evidence discipline, and prevent inconsistent narratives across departments.
This document is not administrative paperwork. It is a compliance control tool that keeps the response grounded in verifiable evidence rather than interpretation.
A well-structured investigation brief typically includes:
- the accreditation requirement or standard referenced
- the finding language exactly as written by reviewers
- the scope of the issue (program, campus, modality, or student population)
- the timeframe relevant to the finding
- the evidence reviewers examined
- the internal evidence map identifying existing and missing documentation
- the documented root cause supported by evidence
- any immediate correction actions taken
- systemic corrective actions with responsible owners and deadlines
- verification evidence demonstrating implementation
- an effectiveness monitoring plan
When institutions consistently prepare this brief for every finding, they move from reactive compliance to managed accreditation compliance systems, which reviewers generally view as a sign of institutional maturity.
Common Mistakes in Accreditation Non-Compliance Responses
Many institutions repeat the same mistakes when responding to accreditation findings. These mistakes often weaken otherwise valid responses.
One common mistake is arguing with the finding instead of investigating it. Clarifications may be appropriate if the record is incorrect, but clarification alone rarely replaces evidence of compliance.
Another mistake is treating training as the default corrective action. Training can support compliance, but training alone rarely creates sustainable control. Reviewers expect to see monitoring systems, accountability structures, and verifiable documentation.
A third mistake is correcting only the specific program or location that triggered the finding while leaving the broader institutional system unchanged. If the requirement applies institution-wide, reviewers typically expect corrective actions that address institutional processes.
A final mistake is skipping the effectiveness check. Without a monitoring plan demonstrating that the corrective action worked over time, responses often read as intention rather than verified correction.
What Accreditation Reviewers Look For

Accreditation reviewers typically evaluate whether institutions can demonstrate a clear and credible compliance process.
Reviewers look for a transparent connection between:
- the accreditation requirement
- the finding or deficiency identified
- the documented root cause
- the corrective action plan
- the evidence of implementation
They also evaluate whether compliance practices are consistent across campuses, programs, and delivery modalities. Inconsistent implementation is one of the most common reasons institutions receive repeated findings during accreditation cycles.
Institutional maturity also matters. Mature institutions can clearly explain:
- who owns compliance responsibilities
- how compliance is monitored internally
- how documentation and evidence are controlled
Finally, reviewers look for institutional integrity. When institutions accurately acknowledge compliance gaps, implement realistic corrective actions, and provide transparent monitoring plans, they tend to build credibility with review teams.
International Considerations for Accreditation Compliance
The investigation principles described in this article apply across most quality assurance and accreditation systems worldwide. While terminology and reporting formats vary, the core elements remain consistent: requirements, evidence, evaluation, and improvement.
What differs internationally is typically:
- terminology used by quality assurance agencies
- documentation formats and reporting structures
- review timelines and response procedures
- the relationship between accreditors, ministries, and regulatory authorities
Institutions operating internationally or delivering cross-border education should maintain a consistent internal investigation process while adapting reporting formats to the relevant accreditor or regulator.
Particular attention should be given to evidence equivalency, since documentation standards and expectations can vary between jurisdictions.
When in doubt, institutions should prepare investigation documentation as if it may be reviewed by an independent external auditor. In most cases, documentation prepared to that standard will meet or exceed typical accreditation expectations.
Frequently Asked Questions About Accreditation Non-Compliance
What is non-compliance in an accreditation context?
Accreditation non-compliance is a documented gap where an institution’s practice or supporting evidence does not meet a stated accreditation requirement or standard. The terminology may vary between accreditors, but the core meaning is the same: the institution must demonstrate compliance through evidence and implement corrective actions that reduce the risk of recurrence.
Do institutions have to agree with an accreditation finding?
No. Institutions can provide evidence-based clarification if they believe the finding is based on incomplete or inaccurate information. However, even when challenging a finding, the institution must still demonstrate compliance with the requirement. Maintaining a contingency correction plan is often recommended in case the finding ultimately stands.
What is the difference between correction and corrective action in accreditation?
A correction fixes a specific instance of non-compliance, such as updating missing documentation or correcting an error.
A corrective action addresses the underlying cause of the issue so it does not recur. Corrective actions typically involve changes to institutional processes, governance oversight, monitoring systems, or documentation controls.
Accreditors typically place greater emphasis on corrective actions because they demonstrate long-term compliance control.
How do institutions conduct root cause analysis for accreditation findings?
Effective root cause analysis requires evidence-based investigation rather than assumptions. Institutions should identify the systemic factors that allowed the non-compliance to occur and connect those factors directly to corrective actions.
If the analysis produces vague conclusions such as “communication problems” without supporting evidence, ownership, or monitoring controls, the response will likely be viewed as weak by accreditation reviewers.
What evidence should be included in a corrective action response?
Strong accreditation corrective action responses include verifiable, dated, and requirement-aligned evidence. This often includes approved policies with version history, governance or committee meeting documentation, audit logs or internal review results, controlled training completion records, updated catalogs or institutional publications, and monitoring reports demonstrating sustained compliance.
How can institutions demonstrate effectiveness if a corrective action is new?
When a corrective action has recently been implemented, institutions should provide clear implementation evidence and define an effectiveness monitoring plan. This plan should explain who will review monitoring results, when evaluations will occur, which indicators will confirm success, and what conditions would trigger additional corrective action. Accreditation reviewers generally prefer realistic monitoring plans supported by early evidence rather than overly confident claims without verification.
Why do accreditation findings repeat across review cycles?
Findings often repeat when institutions correct individual instances of non-compliance but fail to address the broader institutional system.
Without changes to governance structures, ownership of requirements, monitoring processes, and evidence controls, the same underlying issue can reappear during future accreditation reviews under different standards.
Can Accreditation Expert Consulting assist with non-compliance investigations?
Yes. Accreditation Expert Consulting (AEC) assists institutions with structured non-compliance investigations, root cause analysis, evidence organization, and corrective action planning. The goal is to help institutions produce reviewer-ready documentation that clearly connects requirements, findings, corrective actions, and implementation evidence. Support is typically most effective when engaged early, before response deadlines create rushed or incomplete compliance narratives.
Next Steps
If your institution has an active accreditation non-compliance finding or expects a corrective action response, treat it like a controlled investigation rather than a writing assignment. Start by freezing the finding language, mapping evidence, and documenting the root cause with supporting proof. Then design corrective actions that strengthen institutional systems, assign accountable owners, and define verification and effectiveness checks that can be consistently executed.
Once the internal investigation is structured and evidence-based, the external response becomes clearer, faster, and far more defensible.
Accreditation Expert Consulting (AEC) can support your team with a structured investigation workflow, evidence indexing and organization, corrective action plan development, and readiness validation so your response is coherent, complete, and reviewer-ready.
About the Author
Accreditation Expert Consulting (AEC) provides practical, fact-based guidance for higher education accreditation, reaccreditation and reaffirmation, accreditation compliance responses, corrective action planning, state authorization and approval, and institutional readiness work.
AEC supports institutional leaders and operators with evidence organization, standards alignment, and documentation strategies designed to stand up to peer review.

Disclaimer
This article is provided for general informational purposes and does not constitute legal advice. Accreditation and regulatory requirements vary by accreditor, jurisdiction, institution type, and scope of operations. Institutions should confirm expectations using official accreditor and regulatory guidance and consult qualified legal or compliance professionals when necessary.



